Extended Pivotal Trial Results for the MiniMed 780G system with the newest Guardian™ 4 sensor Demonstrate Sustained Improvements in Time in Range with Less User Interaction
Results were presented at the Advanced Technologies & Treatments for Diabetes (ATTD) Conference
Medtronic reported study results from the extended study phase of its MiniMed™ 780G system[i] pivotal trial with new data using its no-calibration, non-adjunctive Guardian™ 4 sensor[ii]. The data demonstrated sustained improvements in glycemic outcomes that met or exceeded consensus guideline recommendations of 70 percent Time in Range with less user interaction and fewer fingerstick calibrations.
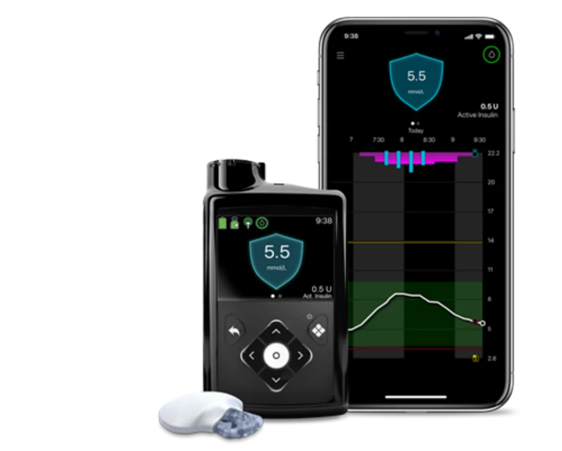
The advanced hybrid closed loop (AHCL) pivotal study started in 2019. At the conclusion of the pivotal study in 2021, participants were provided the opportunity to stay on product as part of an extended study phase and continue to monitor their progress. The data presented from the extension study phase includes 176 individuals aged 7 – 75 years old.
It showed overall Time in Range (70-180 mg/dL) of 72.8%. Adults saw an average Time in Range of 76.3% and children aged 7 – 17 saw an average Time in Range of 70.7%. There were no episodes of diabetic ketoacidosis (DKA) or severe hypoglycemia. Additionally, the average time spent in advanced hybrid closed loop mode was over 90% across all age groups.
|
Interim Analysis Outcomes of MiniMed™ 780G with Guardian™ 4 Sensor Use |
|||
|
|
Overall (n=176) |
Pediatrics, 7 – 17 (n=109) |
Adults, 18-75 (n=67) |
|
< 54 mg/dL |
0.4% |
0.4% |
0.3% |
|
<70 mg/dL |
1.9% |
2.2% |
1.6% |
|
72.8% |
70.7% |
76.3% |
|
> 180 mg/dL |
25.2% |
27.1% |
22.1% |
|
>250 mg/dL |
6.1% |
7.3% |
4.3% |
The transition to the latest Guardian™ 4 sensor, from the previous generation Guardian™ sensor 3 meant that extended study participants no longer had to calibrate their sensor a minimum two times per day. Even with fewer calibrations, individuals safely achieved glycemic targets similar to that observed in the pivotal trial of the AHCL system with Guardian™ sensor 3.
The MiniMed™ 780G system is now available in over 40 countries around the world and is currently being reviewed by the Food and Drug Administration (FDA) for approval in the U.S.
About the MiniMedTM 780G system
The MiniMed™ 780G system is the most advanced insulin pump system from Medtronic, currently approved for the treatment of type 1 diabetes in people age 7 to 80 years. The MiniMed™ 780G system's SmartGuard algorithm (also referred to as the advanced hybrid closed-loop algorithm) automates the delivery of insulin every five minutes — personalizing these doses to auto-correct highs and lows 24 hours a day based on CGM readings[iii] [iv]. The system enables the personalization of glucose goals with an adjustable target setting as low as 100 mg/dL (5.5 mmol/L).
About the Diabetes Business at Medtronic (www.medtronicdiabetes.com)
Medtronic is working together with the global community to change the way people manage diabetes. The company aims to transform diabetes care by expanding access, integrating care and improving outcomes, so people living with diabetes can enjoy greater freedom and better health.
For more information, contact:
Contacts:
Kendra Cassillo Ryan Weispfenning
Public Relations Investor Relations
+1-818-576-5611 +1-763-505-4626
[i] MiniMed 780G System and Guardian™ 4 sensors are CE marked only and not commercially available or approved in the U.S.
[ii] Medtronic data on file.
[iii] Carlson, A.L. et al. Safety and glycemic outcomes during the MiniMed™ Advanced Hybrid Closed-Loop system pivotal trial in adolescents and adults with type 1 diabetes. Diab Tech Ther 2021; in press.
