First commercial North American procedure using Medtronic Hugo™ robotic-assisted surgery system at Toronto General Hospital
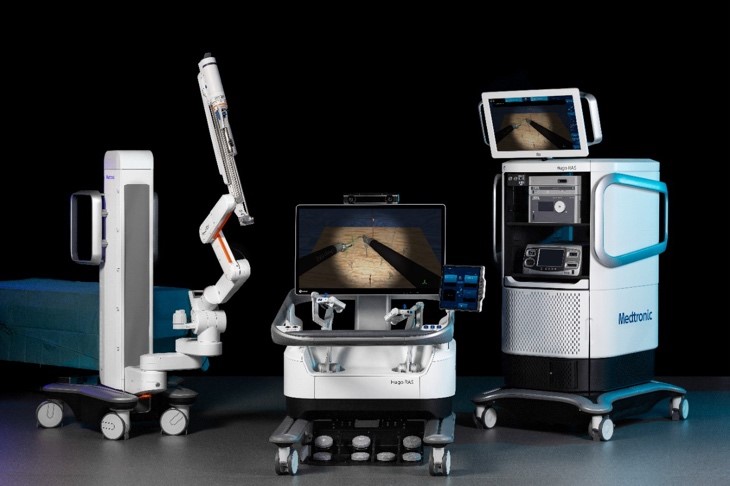
Medtronic Canada is pleased to share that the first commercial North American surgical procedure using the HugoTM robotic-assisted surgery (RAS) system was completed at Toronto General Hospital (TGH), part of University Health Network (UHN).
Surgeons performed a partial nephrectomy with the Hugo RAS system on April 26, 2023, at TGH, one of Canada's Top 40 Research Hospitals.
"It’s exciting to see the continuous advancement of RAS in our institution because patients ultimately benefit from state-of-the-art, minimally invasive procedures,” says Dr. Antonio Finelli, head of the urology division at UHN. “It also means our surgical teams are on the leading edge of technology globally.”
RAS benefits both the patient experience and hospital systems due to faster recovery, fewer complications, and reduced length of stay compared to open procedures. [i],[ii],[iii]
The Hugo RAS system is modular, combining wristed instruments and 3D visualization with dedicated support teams specializing in robotics program optimization, service, and training. Hugo is compatible with Touch Surgery™ Enterprise, a first-of-its-kind AI-powered platform that simplifies how surgeons record, store, share and analyze surgical video and data.†
In Canada, the Hugo RAS system is indicated for use in urologic, gynecologic laparoscopic, and general laparoscopic surgical procedures, approximately 95% of all laparoscopic RAS currently performed in the country.[iv]
“We heartily congratulate the UHN team on being the first Canadian centre to use the Hugo RAS system in a clinical procedure, as well as being home to the first commercial system in North America,” says Sheri Dodd, president of Medtronic Canada. “We’re looking forward to partnering with Canadian health systems to help address efficient and effective delivery of care through world-class robotic surgery programs. We’re here to support clinician adoption of RAS care so that together we can contribute to improved patient experiences and outcomes.
The Hugo RAS system is not cleared or approved in all markets. Regulatory requirements of individual countries and regions will determine approval, clearance, or market availability. In the U.S., the Hugo RAS system is an investigational device not for sale.
For more media details, please visit CanadaNews.medtronic.com and follow us on Twitter and LinkedIn.
For more information, contact:
Roxane Bélanger, Ryan Weispfenning,
Public Relations Investor Relations
+1-416-553-0479 +1-763-505-4626
†Touch Surgery™ Enterprise is not intended to direct surgery, or aid in diagnosis or treatment of a disease or condition.
[ii] Tiwari MM, Reynoso JF, High R, Tsang AW, Oleynikov D. Safety, efficacy, and cost effectiveness of common laparoscopic procedures. Surg Endosc. 2011;25(4):1127-1135.
[iii] Roumm AR, Pizzi L, Goldfarb NI, Cohn H. Minimally invasive: minimally reimbursed? An examination of six laparoscopic surgical procedures. Surg Innovation. 2005;12(3):261–287.
[iv] Based on Medtronic report, FY20 market model: procedural volume data.
