| Su | Mo | Tu | We | Th | Fr | Sa |
|---|---|---|---|---|---|---|
| Su | Mo | Tu | We | Th | Fr | Sa |
|---|---|---|---|---|---|---|
-
Aug 25, 2023
Inceptiv automatically and instantly adjusts therapy based on signals from the spinal cord DUBLIN, Aug. 25, 2023 /PRNewswire/ -- Medtronic plc (NYSE:MDT), a global leader in healthcare technology,...
-
Oct 26, 2022
New Medtronic Neurovascular Co-Lab™ Platform drives collaboration and connections among start-ups, physicians, and institutions; aims to improve stroke treatment and access for patients...
-
Jul 14, 2022
Powered by a database of over 10,000 surgical cases, Medtronic continues to evolve the UNiD™ platform to help surgeons plan procedures, personalize care for their patients and predict...
-
May 13, 2022
Acquisition adds innovative bioabsorbable steroid-eluting sinus implants to ENT portfolio DUBLIN and MENLO PARK, Calif., May 13, 2022 /PRNewswire/ -- Medtronic plc (NYSE: MDT), a global leader in...
-
Mar 24, 2022
TITAN 2 pivotal study to evaluate implantable tibial neuromodulation (TNM) device to help expand patient access to advanced therapy DUBLIN, March 24, 2022 /PRNewswire/ -- Medtronic plc (NYSE:...
-
Mar 23, 2022
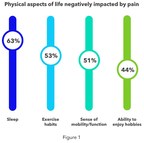
Medtronic and Harris Poll survey finds 4 in 5 report chronic back or leg pain is worse or unimproved since the COVID-19 pandemic began DUBLIN, March 23, 2022 /PRNewswire/ -- A new survey...
-
Feb 28, 2022Single-use device treats Eustachian tube dysfunction during office procedures
Medtronic plc (NYSE: MDT), a global leader in healthcare technology, today announced the launch of the NuVent™ Eustachian tube dilation balloon, which has been cleared by the U.S. Food and Drug...
-
Feb 22, 2022- New InterStim X™ system provides 10 to 15 years of battery life without the need to recharge
Medtronic plc (NYSE:MDT), a global leader in healthcare technology, today announced it received approval from the U.S. Food and Drug Administration (FDA) for InterStim X™ ― the next generation...
-
Jan 24, 2022Studies show significant reduction in pain and improved quality of life for patients treated with spinal cord stimulation compared to medication management alone
Medtronic plc (NYSE:MDT), a global leader in healthcare technology, today announced it has received U.S. Food and Drug Administration approval of its Intellis™ rechargeable neurostimulator and...
-
Jan 14, 2022
DTM™ SCS endurance therapy enables long-lasting recharge-free performance on Vanta™ and 5-minute recharge on Intellis™ neurostimulators DUBLIN, Jan. 14, 2022 /PRNewswire/ -- Medtronic plc...