| Su | Mo | Tu | We | Th | Fr | Sa |
|---|---|---|---|---|---|---|
| Su | Mo | Tu | We | Th | Fr | Sa |
|---|---|---|---|---|---|---|
-
Mar 23, 2026
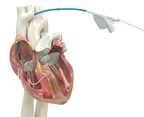
Built for high reliability, world's only lumenless defibrillation lead approved for placement in the left bundle branch area GALWAY, Ireland, March 23, 2026 /PRNewswire/ -- Medtronic plc (NYSE:...
-
Feb 3, 2026
Deal demonstrates Medtronic's commitment to expanding pipeline through strategic investments and targeted acquisitions Exercising option to acquire will bolster Medtronic's interventional...
-
Apr 25, 2025
Adding to the Medtronic portfolio of catheter-based lead solutions, the novel OmniaSecure defibrillation lead allows for precise delivery and placement in the right ventricle Heart Rhythm 2025:...
-
Jan 13, 2025
European approval introduces the world's only closed-loop DBS system with real-time, self-adjusting brain stimulation for people with Parkinson's disease GALWAY, Ireland, Jan. 13, 2025...
-
May 17, 2024
Global LEADR clinical trial meets safety and effectiveness objectives; results presented at Heart Rhythm 2024 and simultaneously published in Heart Rhythm DUBLIN and BOSTON, May 17, 2024...
-
Apr 8, 2024
EHRA late-breaking data: Results highlight efficacy, safety, and durability of the novel PFA catheter that is fully integrated with Affera™ Mapping and Ablation System DUBLIN and BERLIN, April...
-
Aug 28, 2022Medtronic Extravascular ICD meets global pivotal clinical trial's safety and effectiveness endpoints
Late-breaking data presented at ESC Congress 2022 and simultaneously published in The New England Journal of Medicine confirms implant procedure safety and defibrillation success DUBLIN and BARCELONA,
-
Jan 21, 2022- Multi-year partnerships to serve over 60,000 underserved and underrepresented K-12 students in Minnesota, Northern California and Puerto Rico
The Medtronic Foundation today announced 13 new partnerships with leading organizations serving underrepresented and underserved K-12 students in Minnesota, Northern California and Puerto Rico....
-
Nov 15, 2021Latest approval for PillCam™ further enables patients to receive gastrointestinal care in the comfort of their own home
Medtronic plc (NYSE:MDT), a global leader in healthcare technology, today announced that the U.S. Food and Drug Administration (FDA) has granted 510(k) clearance for its PillCam™ Small Bowel 3...
-
Sep 30, 2021Industry Leading Portfolio of Integrated Solutions Sets a New Standard for Minimally Invasive Spine Procedures, Beginning with MIS+ TLIF
Medtronic plc (NYSE: MDT), the global leader in medical technology, today announced the latest additions to its minimally invasive spine surgery ecosystem, making it the only company to combine...