| Su | Mo | Tu | We | Th | Fr | Sa |
|---|---|---|---|---|---|---|
| Su | Mo | Tu | We | Th | Fr | Sa |
|---|---|---|---|---|---|---|
-
Jan 8, 2024
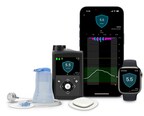
With CE Mark approval, the benefits of the MiniMed™ 780G system are now available with a new sensor that takes less than 10 seconds to insert1 DUBLIN, Jan. 8, 2024 /PRNewswire/ -- Medtronic plc...
-
Jan 5, 2024
New pacemakers offer 40% more battery life,1 extend Medtronic legacy of pacing leadership DUBLIN, Jan. 5, 2024 /PRNewswire/ -- Medtronic plc (NYSE: MDT), a global leader in healthcare technology,...
-
Dec 18, 2023
Medtronic plc (NYSE:MDT), a global leader in healthcare technology, today announced it will participate in the 42nd annual J.P. Morgan healthcare conference on Monday, January 8, 2024. Geoff...
-
Dec 13, 2023
Safe, efficient, and effective treatment for both paroxysmal and persistent atrial fibrillation DUBLIN, Dec. 13, 2023 /PRNewswire/ -- Medtronic plc (NYSE: MDT), a global leader in healthcare...
-
Dec 11, 2023
Agreement expands partnership between Medtronic and Cosmo Pharmaceuticals, set to transform endoscopy with cutting-edge AI technology DUBLIN, Dec. 11, 2023 /PRNewswire/ -- Medtronic plc (NYSE:...
-
Dec 7, 2023
The board of directors of Medtronic plc (NYSE:MDT) on Thursday, December 7, 2023, approved the company's cash dividend for the third quarter of fiscal year 2024 of $0.69 per ordinary share. This...
-
Nov 21, 2023
Solid execution results in mid-single digit revenue growth, driven by broad-based strength across multiple businesses and geographies; Major innovative product approvals; Raises fiscal year...
-
Nov 17, 2023
The Symplicity™ blood pressure procedure offers patients a new adjunct approach to lowering blood pressure Approval is the culmination of ten years of clinical research and development of the...
-
Nov 13, 2023
According to a new survey from Medtronic and Morning Consult, respondents recognize potential of AI to enable earlier diagnosis and improve access to care DUBLIN, Nov. 13, 2023 /PRNewswire/ --...
-
Nov 7, 2023
Medtronic plc (NYSE: MDT), a global leader in healthcare technology, today announced that it will report financial results for its second quarter of fiscal year 2024 on Tuesday, November 21, 2023....