Q4 earnings: three things to know
Medtronic finishes the fiscal year with momentum
Medtronic announced financial results for the fourth quarter and full fiscal year on Thursday. Here are three things to know:

A strong finish to the fiscal year
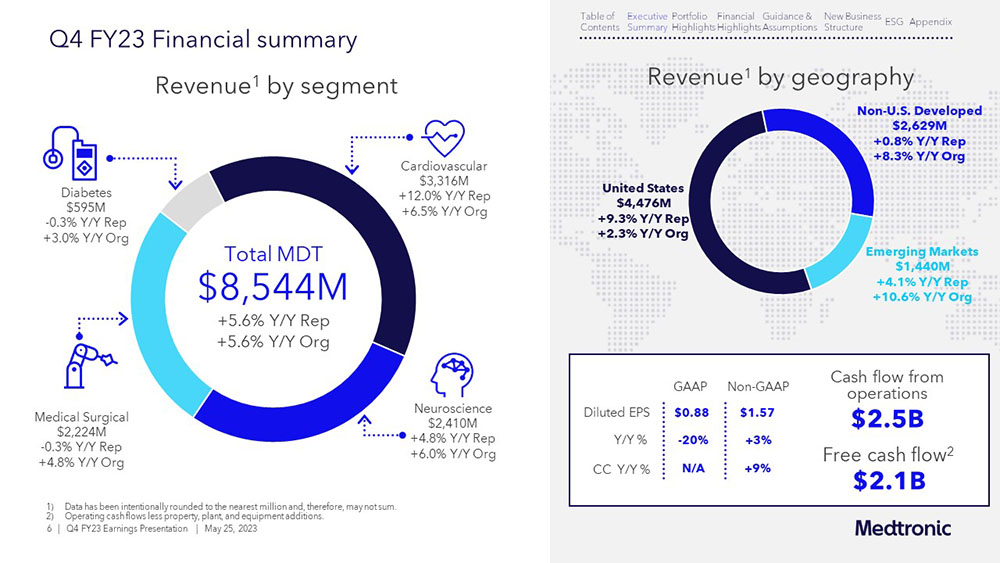
The performance of several businesses – including many in the cardiovascular, neuroscience, and medical surgical portfolios – boosted sales during the fourth quarter. An increase in medical procedures, supply chain improvements, and introductions of innovative products helped, too.
Pipeline momentum
The Medtronic pipeline is strong and gaining momentum. The MiniMed™ 780G system with Guardian™ 4 sensor for people living with diabetes recently received U.S. Food and Drug Administration approval. The Affera™ mapping and ablation system – including our Sphere-9™ catheter – received CE Mark in March. And innovative technologies like our Micra™ leadless pacemaker continue to reach more patients.
Transforming for growth and looking ahead
Behind the scenes, Medtronic is working to deliver durable growth by investing in the right opportunities and executing well. The company increased its dividend for the 46th consecutive year and announced its FY 2024 guidance.
L009-05242023
Related content