When fit happens (by good design)
How good ergonomic design shapes the development of healthcare technology at Medtronic
Surgeons help patients by restoring their health. But who is responsible for keeping surgeons healthy?
Good ergonomic design of healthcare technology — such as surgical robots and advanced instrumentation — goes a long way toward protecting the health of clinicians. Without it, they run the risk of work-related musculoskeletal disorders (WMSDs) or injuries such as carpal tunnel syndrome and tendonitis.
It’s a serious concern — One study found that about 20 percent of surgeons that perform minimally invasive surgeries are at risk for WMSDs.
At Medtronic, teams of engineers focus on making sure good ergonomic design is baked into the development of all our life-saving medical devices. It’s an intentional process that often takes years and requires deep collaboration with skilled surgeons.
“Surgeon comfort is becoming more top of mind,” said Dr. Erik Wilson, director of bariatric surgery at Memorial Herman-Texas Medical Center. “Surgeons are expensive to train, and it may take years of training to become an expert. From there, you can expect a 20 to 25 year run. If we can add 10 to 20 years to that through better ergonomics, then there would be fewer stress injuries forcing surgeons to quit.”
Putting people at the center of design
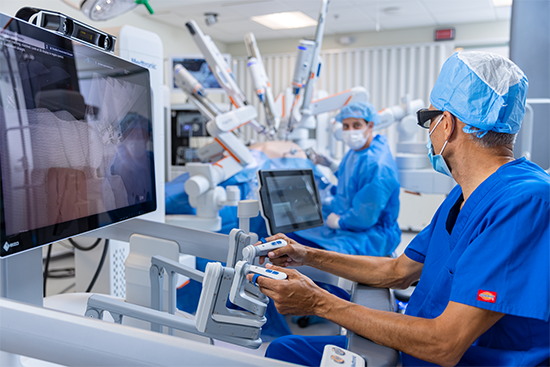
A great example of good ergonomic design in action is our new soft-tissue surgical robot.
Clinical engineers, human factors engineers, and industrial designers started the design process by analyzing operating room workflows and asking questions like, “How many people will be in the room at one time?” “How do clinicians need to access the patient?” and “Where are the people and equipment in the room located?”
They then had to determine what clinicians needed most to perform a successful minimally invasive surgery and sought input from hundreds of highly skilled surgeons. Not surprisingly, being comfortable was at the top of the wish list for surgeons, many of whom often stand for hours during complicated procedures.
Their feedback ultimately led to the development of a robotic-assisted surgical system with an open console where surgeons could work in a seated position and maintain good line of sight with the patient and OR staff.
Creating healthcare technology that puts people — in this case surgeons — at the forefront of the design process is known as human-centered design.

Joe Cesa, who leads the User Centered Design Team for the Surgical business at Medtronic, said the team that designed the system had to accommodate users between 4’11" and 6’2" (95% of all people). Consideration was also given to whether surgeons were left-handed or right-handed, and whether they had physical impairments like hearing or vision loss.
"It all comes down to flexibility and being able to adjust the system and configure it in a way that best fits what clinicians need,” Cesa said. “The 3D surgeon glasses are designed to fit over prescription glasses. The armrests can go up and down. The foot pedals can move in and out. The monitors move up and down. The arm carts have large handles that you can grab up or down. Ergonomics factored into every aspect of those design choices.”
It’s not just about fit
How healthcare technology feels to a user is incredibly important. But it’s not the only aspect of good ergonomic design. There’s also cognitive ergonomics.
Within our industry, that means designing healthcare technology that has clear user interfaces and can effectively relay information back to the user to make sure they can use it safely and effectively.
For our robotic-assisted surgical system, engineers had to create controls that were easy to find and clearly labeled. They considered how colors, font sizes, and even sounds, relayed information to users.
“When you see blinking yellow lights, you automatically think, ‘Something went wrong.’ And a red light? That’s even worse,” Cesa said. “As designers and engineers, it’s our job to think about the clearest and simplest way for the device to communicate with users and that the user’s beliefs align with that communication. That’s the main objective of cognitive ergonomics.”
Cesa said his human factors team analyzed more than 12,000 data points associated with using system. That’s a huge amount of data to review. But the work is necessary to identify whether users might struggle to operate the system.
“Humans are not designed engineered systems,” Cesa said. “We are completely unpredictable. Trying to design something that behaves the way a human would think is a really hard thing. But that's why our team exists — we try to anticipate how are people are going to respond to our technology based on knowledge of human behavior.”
The Medtronic Hugo™ RAS system is commercially available in certain geographies. Regulatory requirements of individual countries and regions will determine approval, clearance, or market availability. In the EU, the Hugo™ RAS system is CE marked. In the U.S., the Hugo™ system is an investigational device not for sale. Touch Surgery™ Enterprise is not intended to direct surgery, or aid in diagnosis or treatment of a disease or condition.
L001-06232023
Related content


