FDA Approves “New Era” DBS Device
Getting insights into the most complicated machine known to humankind – the human brain – may be getting easier.
July 7, 2020: Getting insights into the most complicated machine known to humankind – the human brain – may be getting easier.
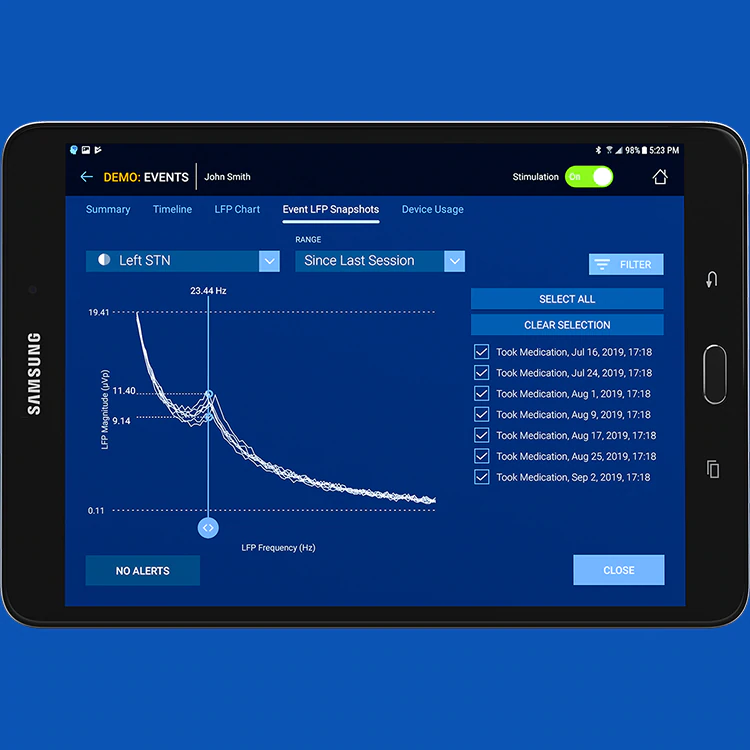
The U.S. Food and Drug Administration (FDA) has approved a new deep brain stimulation (DBS) device from Medtronic called Percept™ PC with BrainSense™ technology, which does something no other device of its kind can do. This new device is the first and only DBS neurostimulation system with the ability to chronically capture and record brain signals while delivering therapy to patients with neurologic disorders associated with Parkinson’s disease, essential tremor, dystonia*, epilepsy or obsessive-compulsive disorder (OCD*).
“BrainSensing for DBS means that for the first time we can record signals directly from the brain,” said Mark Richardson, M.D., Ph.D., Director of Functional Neurosurgery at Massachusetts General Hospital. “This means we now have a read out of what is happening during DBS, to see how brain activity changes with stimulation.”
Mayo Clinic in Rochester, MN will be the first in the United States to implant the new PerceptTM PC device. “Our goal is for patients to regain independence, and we know that DBS can significantly improve motor function in people with Parkinson’s disease compared to standard medication alone,” said Bryan Klassen, M.D., neurologist, Mayo Clinic. “We can now more precisely tailor therapy to the individual needs of each patient based on data from neuronal activity.”
The FDA first approved DBS to treat symptoms of essential tremor and Parkinsonian tremor in 1997. DBS therapy uses a surgically implanted medical device, similar to a cardiac pacemaker, to deliver electrical stimulation to precisely targeted areas of the brain as adjunctive treatment for several neurological disorders.
Scott Stanslaski, a 25-year Medtronic engineer, was among a handful of researchers that saw the PerceptTM PC device from beginning to end.
“Being able to sense and separate brain signals amid the other noise was by far our biggest challenge,” Stanslaski said. “One of the difficulties with sensing brain signals is that they are very small compared to the stimulation therapy that we deliver – they are about a million times smaller1.”
“BrainSensing for DBS means that for the first time we can record signals directly from the brain,” said Mark Richardson, M.D., Ph.D., Director of Functional Neurosurgery at Massachusetts General Hospital. “This means we now have a read out of what is happening during DBS, to see how brain activity changes with stimulation.”
Mayo Clinic in Rochester, MN will be the first in the United States to implant the new PerceptTM PC device. “Our goal is for patients to regain independence, and we know that DBS can significantly improve motor function in people with Parkinson’s disease compared to standard medication alone,” said Bryan Klassen, M.D., neurologist, Mayo Clinic. “We can now more precisely tailor therapy to the individual needs of each patient based on data from neuronal activity.”
The FDA first approved DBS to treat symptoms of essential tremor and Parkinsonian tremor in 1997. DBS therapy uses a surgically implanted medical device, similar to a cardiac pacemaker, to deliver electrical stimulation to precisely targeted areas of the brain as adjunctive treatment for several neurological disorders.
Scott Stanslaski, a 25-year Medtronic engineer, was among a handful of researchers that saw the PerceptTM PC device from beginning to end.
“Being able to sense and separate brain signals amid the other noise was by far our biggest challenge,” Stanslaski said. “One of the difficulties with sensing brain signals is that they are very small compared to the stimulation therapy that we deliver – they are about a million times smaller1.”
Important Information
* Humanitarian Device: The effectiveness of this device for the treatment of dystonia and obsessive-compulsive disorder has not been demonstrated.
† Medtronic DBS systems are MR Conditional and safe in the MR environment as long as certain conditions are met. If the conditions are not met, a significant risk is tissue lesions from component heating, especially at the lead electrodes, resulting in serious and permanent injury including coma, paralysis, or death. Refer to the MRI Guidelines for Medtronic Deep Brain Stimulation Systems for a complete list of conditions: http://professional.medtronic.com/mri.
‡ For median energy use in DBS for PD patients, with equivalent settings and no BrainSense technology usage
References
L001-06252020, UC202103325EN