| Su | Mo | Tu | We | Th | Fr | Sa |
|---|---|---|---|---|---|---|
| Su | Mo | Tu | We | Th | Fr | Sa |
|---|---|---|---|---|---|---|
-
Mar 23, 2026
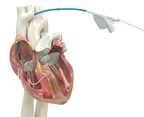
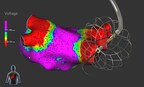
Built for high reliability, world's only lumenless defibrillation lead approved for placement in the left bundle branch area GALWAY, Ireland, March 23, 2026 /PRNewswire/ -- Medtronic plc (NYSE:...
-
Feb 3, 2026
Deal demonstrates Medtronic's commitment to expanding pipeline through strategic investments and targeted acquisitions Exercising option to acquire will bolster Medtronic's interventional...
-
Apr 25, 2025
Adding to the Medtronic portfolio of catheter-based lead solutions, the novel OmniaSecure defibrillation lead allows for precise delivery and placement in the right ventricle Heart Rhythm 2025:...
-
Jan 13, 2025
European approval introduces the world's only closed-loop DBS system with real-time, self-adjusting brain stimulation for people with Parkinson's disease GALWAY, Ireland, Jan. 13, 2025...
-
May 17, 2024
Global LEADR clinical trial meets safety and effectiveness objectives; results presented at Heart Rhythm 2024 and simultaneously published in Heart Rhythm DUBLIN and BOSTON, May 17, 2024...
-
Apr 8, 2024
EHRA late-breaking data: Results highlight efficacy, safety, and durability of the novel PFA catheter that is fully integrated with Affera™ Mapping and Ablation System DUBLIN and BERLIN, April...
-
Aug 28, 2022Medtronic Extravascular ICD meets global pivotal clinical trial's safety and effectiveness endpoints
Late-breaking data presented at ESC Congress 2022 and simultaneously published in The New England Journal of Medicine confirms implant procedure safety and defibrillation success DUBLIN and BARCELONA,
-
Jan 21, 2022- Multi-year partnerships to serve over 60,000 underserved and underrepresented K-12 students in Minnesota, Northern California and Puerto Rico
The Medtronic Foundation today announced 13 new partnerships with leading organizations serving underrepresented and underserved K-12 students in Minnesota, Northern California and Puerto Rico....
-
Nov 15, 2021Latest approval for PillCam™ further enables patients to receive gastrointestinal care in the comfort of their own home
Medtronic plc (NYSE:MDT), a global leader in healthcare technology, today announced that the U.S. Food and Drug Administration (FDA) has granted 510(k) clearance for its PillCam™ Small Bowel 3...
-
Sep 30, 2021Industry Leading Portfolio of Integrated Solutions Sets a New Standard for Minimally Invasive Spine Procedures, Beginning with MIS+ TLIF
Medtronic plc (NYSE: MDT), the global leader in medical technology, today announced the latest additions to its minimally invasive spine surgery ecosystem, making it the only company to combine...
-
Sep 28, 2021- Portfolio Features First Catheter Specifically Designed to Allow Neurointerventionalists Radial Artery Access, Delivers Advantages to Patients Treated for Stroke, Brain Aneurysms, Other Neurovascular Conditions
Medtronic plc (NYSE:MDT), the global leader in medical technology, today announced it has received CE Mark approval for its radial artery access portfolio, which includes the Rist™ 079 Radial...
-
Sep 7, 2021
Results Demonstrate Feasibility of Bronchoscopic Microwave Ablation for Certain Patients with Malignant Lung Nodules DUBLIN, Sept. 7, 2021 /PRNewswire/ -- Medtronic plc (NYSE:MDT), the global...
-
Aug 24, 2021Evolut™ FX TAVR System Adds Innovative Features to Enhance Ease-of-Use and Predictable Valve Deployment
Medtronic plc (NYSE: MDT), the global leader in medical technology, today announced U.S. Food and Drug Administration (FDA) approval of its newest-generation, self-expanding transcatheter aortic...
-
Jun 7, 2021The SenSight™ Directional Lead System Was Designed to Enable Precision, Personalization and Enhanced Patient Comfort
Medtronic plc (NYSE:MDT), the global leader in medical technology, today announced the U.S. Food and Drug Administration (FDA) approval and first U.S. implants of the SenSight™ Directional Lead...
-
May 10, 2021SonarMed™ Airway Monitoring System Continuously Checks for Endotracheal Tube Obstruction and Position for Neonates and Infants Providing Immediate, Actionable Intelligence for Clinicians
Medtronic plc (NYSE:MDT), the global leader in medical technology, today announced the U.S. commercial launch of the SonarMed™ airway monitoring system. The first and only device of its kind,...
-
Apr 26, 2021SyncAR™ Technology Uniquely Integrates with StealthStation™ S8 Surgical Navigation System and Expands the Use of Augmented Reality Into Operating Rooms to Enhance Precision and Efficiency
Medtronic plc (NYSE:MDT), the global leader in medical technology, and Surgical Theater today announced a partnership to interface Surgical Theater's SyncAR™ augmented reality (AR) technology...
-
Apr 7, 2021New Partnerships and Research Efforts Prioritized Over the Next Three Years to Address Health Disparities
Medtronic plc (NYSE:MDT), the global leader in medical technology, today announced its ongoing commitment to health equity for people of color living with diabetes. The company committed...
-
Mar 22, 2021
This First-of-its-Kind Directional Lead Combines the Benefits of Directionality with the Power of Sensing DUBLIN, March 22, 2021 /PRNewswire/ -- Medtronic plc (NYSE:MDT), the global leader in...
-
Mar 17, 2021Revised Commercial Labeling to Reflect Outcomes of a Randomized Controlled Trial Demonstrating Superiority of DTM™ Spinal Cord Stimulation (SCS) Compared to Conventional SCS for the Treatment of Chronic Intractable Back Pain
Medtronic plc (NYSE: MDT), the global leader in medical technology, today announced the U.S. Food and Drug Administration (FDA) has approved revised commercial labeling for the Intellis™...
-
Feb 18, 2021
Medtronic plc (NYSE:MDT), the global leader in medical technology, today announced it has received approval from the United States Food and Drug Administration (FDA) for expanded MRI labeling of...
-
Jan 29, 2021Clinical Trial Demonstrates Procedural Efficiencies, Safety, Effectiveness and Non-Inferiority of DiamondTemp System
Medtronic plc (NYSE:MDT), the global leader in medical technology, today announced it has received U.S. Food and Drug Administration (FDA) approval of the DiamondTemp™ Ablation (DTA) system...
-
Jan 14, 2021
ADAPT-PD Global Study Designed to Demonstrate Safety & Efficacy of Automated Therapy Which Responds to Brain Signals in Parkinson's Disease Patients DUBLIN, Jan. 14, 2021 /PRNewswire/ -- Medtronic...
-
Dec 15, 2020The OPuS One Clinical Study is the largest study with the most meaningful clinical results supporting the OsteoCool™ Technology to-date
Medtronic plc (NYSE: MDT) today announced the release of outcomes in the first 100 patients treated with the OsteoCool™ Radiofrequency Ablation (RFA) System in the OPuS One Clinical Study (as...
-
Nov 30, 2020- New Commitment Includes Nearly $4 Million for Health Workers, Mental Health, Food Assistance, and Critical Services in Black, Indigenous and People of Color (BIPOC) Communities
With the pandemic surging across the globe and food insecurity on the rise, Medtronic and the Medtronic Foundation today announced an additional $3.8 million commitment to provide healthcare...
-
Oct 29, 2020New Technologies Address Most Common Challenges During Head and Neck Surgery
Medtronic plc (NYSE:MDT), the global leader in medical technology, today announced it has received U.S. Food and Drug Administration (FDA) 510(k) clearance of the NIM Vital™ nerve monitoring...
-
Oct 20, 2020Study Reports 84% Back Pain Responder Rate and 69% Profound Responder Rate at 12 Months(1)
Medtronic plc (NYSE:MDT), the global leader in medical technology, today announced statistically significant 12-month results from a large, multicenter randomized controlled trial (RCT) that...
-
Oct 9, 2020In Partnership with the Medtronic Foundation, More Than 400,000 KN95 Respirators and 1.1 Million Surgical Masks to be Distributed to Healthcare Facilities Across Continental U.S. and Puerto Rico
Medtronic plc (NYSE:MDT), the global leader in medical technology, today announced that it has donated nearly $1 million worth of personal protective equipment (PPE) to relief organization...
-
Aug 5, 2020First Patient Receives New Sacral Neuromodulation Device Implant at Cleveland Clinic
DUBLIN, Aug. 05, 2020 (GLOBE NEWSWIRE) -- Medtronic plc (NYSE:MDT), the global leader in medical technology, today announced that its recently FDA-approved InterStim™ Micro neurostimulator for...
-
Aug 3, 2020World’s Smallest Rechargeable Bladder/Bowel Control System — InterStim Micro — Features Trusted MRI Technology for Full-Body 1.5 and 3 Tesla MRI Scans
DUBLIN, Aug. 03, 2020 (GLOBE NEWSWIRE) -- Medtronic plc (NYSE:MDT), the global leader in medical technology, today announced it has received approval from the United States Food and Drug...
-
Jul 22, 2020Medtronic Foundation Commits to First-of-Its-Kind National Partnership with the Thurgood Marshall College Fund, and to Minneapolis with Support of the Northside Achievement Zone, People’s Center Clinics & Services, Among Others
MINNEAPOLIS, July 22, 2020 (GLOBE NEWSWIRE) -- The Medtronic Foundation today announced a $16 million commitment to partnerships with organizations working to bring about social justice and...
-
Jun 25, 2020Next-Generation Technology Makes Percept™ the First and Only DBS System with Ability to Capture Patient-Specific Brain Signals
DUBLIN, June 25, 2020 (GLOBE NEWSWIRE) -- Medtronic plc (NYSE:MDT), the global leader in medical technology, today announces it received Food and Drug Administration (FDA) approval for the...
-
Apr 8, 2020FDA Authorizes Use of PB560 in the United States
DUBLIN, April 08, 2020 (GLOBE NEWSWIRE) -- Medtronic plc (NYSE:MDT), the global leader in medical technology, today announced updates regarding its efforts to increase ventilator production around...
-
Mar 18, 2020
DUBLIN, March 18, 2020 (GLOBE NEWSWIRE) -- Medtronic plc (NYSE:MDT), the global leader in medical technology, continues to make progress in increasing ventilator production worldwide. The company...
-
Feb 4, 2020SPYRAL DYSTAL Study to Evaluate the Effects of RDN Using Fewer, Targeted Ablations to Achieve Meaningful Blood Pressure Reductions
DUBLIN, Feb. 04, 2020 (GLOBE NEWSWIRE) -- Medtronic plc (NYSE:MDT) today announced it will begin enrollment in a pilot study evaluating the safety and efficacy of the Symplicity™ Spyral renal...
-
Jan 21, 2020At Three-Months, 80% of Patients Treated with DTM SCS Reported Back Pain Relief of at Least 50%; 63% Reported Profound Back Pain Relief of 80% or Greater1
DUBLIN, Jan. 21, 2020 (GLOBE NEWSWIRE) -- Medtronic plc (NYSE:MDT) today announced three-month results from a large, multicenter randomized controlled trial (RCT) showing statistically significant...
-
Jan 9, 2020This Next-Generation DBS Technology Allows Physicians to Review Patient-Specific Brain Signals, Enabling More Personalized, Data-Driven Neurostimulation Treatment for Patients
DUBLIN, Jan. 09, 2020 (GLOBE NEWSWIRE) -- Medtronic plc (NYSE:MDT) today announced the CE (Conformité Européenne) Mark for Percept™ PC neurostimulator; it is the only Deep Brain Stimulation...
-
Jan 8, 2020Randomized Control Trial Results to be Presented at NANS Jan. 23-26 in Las Vegas; New, Proprietary Differential Target Multiplexed (DTM) Therapy Available on Medtronic Intellis Spinal Cord Stimulation Platform
DUBLIN, Jan. 08, 2020 (GLOBE NEWSWIRE) -- Medtronic plc (NYSE:MDT) announced it has acquired Stimgenics, LLC, a privately-held company based in Bloomington, Illinois, that has pioneered a novel...
-
Oct 30, 2019System Offers at Home Therapy Management for DBS patients Using Patient-Friendly Consumer Technology
DUBLIN and SAN JOSE, Calif., Oct. 30, 2019 (GLOBE NEWSWIRE) -- Medtronic plc (NYSE:MDT) today announced the U.S. launch of its advanced Patient Programmer technology for Deep Brain Stimulation...
-
Jul 22, 2019Technology Has the Potential to Save Crucial Time and Increase Access to Lifesaving Stroke Treatments
DUBLIN and SAN FRANCISCO, July 22, 2019 (GLOBE NEWSWIRE) -- Medtronic plc (NYSE:MDT), a global leader in medical technology, and Viz.ai, the emerging leader in applied artificial intelligence (AI)...
-
Jun 8, 2019
DUBLIN - June 8, 2019 - Medtronic plc (NYSE:MDT), the global leader in medical technology, today announced that it has initiated the company's pivotal trial for its next-generation Guardian(TM)...
-
May 1, 2019Attain Stability(TM) Quad MRI SureScan(TM) Lead Designed for Precise Placement and Stability
DUBLIN - May 1, 2019 - Medtronic plc (NYSE:MDT) announced it has received U.S. Food and Drug Administration (FDA) approval for the Attain Stability(TM) Quad MRI SureScan(TM) left heart lead....
-
Feb 20, 2019With Official Launch and First Implant, Several Health Insurers Now Consider DBS Therapy for Epilepsy a Covered Indication
DUBLIN - February 20, 2019 - Medtronic plc (NYSE:MDT) today announced both the U.S. launch of Deep Brain Stimulation (DBS) for medically-refractory epilepsy and the first commercially implanted...
-
Feb 7, 2019Approval Based on Clinical Data from the PREMIER Trial - New Indication Provides Options for Patients with Small or Medium, Wide-Necked Brain Aneurysms
DUBLIN - February 7, 2019 - Medtronic plc (NYSE:MDT) announced today that it has received U.S. Food and Drug Administration (FDA) approval on an expanded indication for its Pipeline(TM) Flex...
-
Sep 13, 2018
(GLOBE NEWSWIRE via COMTEX) --Company Marks its Eleventh Consecutive Year on the North American Dow Jones Sustainability Index (DJSI) DUBLIN - September 13, 2018 - Medtronic plc (NYSE:MDT) was...
-
Jun 13, 2018FDA Approved, DBS Clinician Programmer Optimizes the Programming Experience By Addressing Day-to-Day Clinical Management Challenges
DUBLIN - June 13, 2018 - Medtronic plc (NYSE:MDT), the global leader in medical technology, today announced that the U.S. Food and Drug Administration (FDA) recently approved its state-of-the art...
-
May 1, 2018Medtronic Receives FDA Approval for Deep Brain Stimulation Therapy for Medically Refractory Epilepsy7-Year Data from the SANTE Trial Showed Median Seizure Frequency Reduction of 75 percent in Patients with Drug-Resistant Epilepsy with Partial-Onset Seizures Receiving DBS Therapy.
DUBLIN - May 1, 2018 - Medtronic plc (NYSE:MDT), the global leader in medical technology, today announced that the U.S. Food and Drug Administration (FDA) has granted premarket approval for Medtronic'
-
Apr 9, 2018Data on Implant Approach, Blood Pressure Management, Length of Stay, and Quality of Life to be Featured During Congress.
DUBLIN - April 9, 2018 - Medtronic plc (NYSE: MDT) today announced a preview of notable clinical data on the HeartWare HVAD® System that will be presented at The International Society of Heart...
-
Feb 28, 2018The Programs Are Expected to Benefit up to 500,000 Patients in Northeast Pennsylvania and Reduce Cost of Care by $100 Million
LEHIGH VALLEY, Pa. and DUBLIN - February 28, 2018 - Today, Medtronic plc (NYSE: MDT), the global leader in medical technology, and Lehigh Valley Health Network (LVHN), an eight-hospital network...
-
Feb 26, 2018FDA Approved, Resolute Onyx 2.0 mm DES Technology Tackles Clinical Challenge of Treating Coronary Artery Disease in Previously Untreatable Patients
DUBLIN - February 26, 2018 - Designed specifically for small vessels, Medtronic plc (NYSE: MDT) today announced the U.S. Food and Drug Administration (FDA) approval and U.S. launch of the Resolute...
-
Nov 13, 2017
DUBLIN - November 13, 2017 - Medtronic plc (NYSE: MDT) today announced several noteworthy presentations of clinical studies and symposia on aortic, peripheral, endoVenous, and vascular...
-
Nov 6, 2017New Neurostimulation System Is a Non-Opioid Alternative for Chronic Pain and Offers Personalized Pain Relief and Advanced Activity Tracking
DUBLIN - November 6, 2017 - Medtronic plc (NYSE:MDT) today announced that it received CE (Conformité Européenne) Mark for the Intellis(TM) platform for both Spinal Cord Stimulation (SCS) and...
-
Oct 6, 2017
DUBLIN - October 6, 2017 - Medtronic plc (NYSE: MDT) today provided an update on the impact to its manufacturing and sales operations as a result of Hurricane Maria. All four of the company's...
-
Sep 18, 2017The Intellis Platform Includes the World's Smallest Implantable Spinal Cord Stimulator and Offers Personalized Pain Relief and Advanced Activity Tracking
DUBLIN - September 18, 2017 - Medtronic plc (NYSE:MDT) today announced FDA approval and U.S. launch of the Intellis(TM) platform for the management of certain types of chronic intractable pain....
-
Aug 28, 2017Published Simultaneously in The Lancet, the Late-Breaking SPYRAL HTN-OFF MED Study at ESC Successfully Isolates RDN Treatment Effect to Show Compelling Efficacy and Safety of Hypertension Procedure
DUBLIN and BARCELONA - August 28, 2017 - Medtronic plc (NYSE:MDT) today announced its intent to move forward with a new renal denervation pivotal trial following positive first results from a...
-
Aug 21, 2017Medtronic Receives CE Mark for Attain Stability(TM) Quad MRI SureScan(TM) Active-Fixation Heart LeadClinical Trial Also Underway in U.S., Canada and Other Geographies
DUBLIN - August 21, 2017 - Medtronic plc (NYSE: MDT) announced it has received CE (Conformité Européene) Mark for the Attain Stability(TM) Quad MRI SureScan(TM) left heart lead. Paired with...
-
Jun 28, 2017Data Presented at the 17th Quadrennial Meeting of the World Society for Stereotactic and Functional Neurosurgery in Berlin
DUBLIN and BERLIN - June 28, 2017 - Medtronic plc (NYSE: MDT) today announced it has received CE (Conformité Européenne) Mark for SureTune(TM)3 software for deep brain stimulation (DBS)....
-
Jun 6, 2017
DUBLIN - June 6, 2017 - Medtronic plc (NYSE: MDT) today announced it has received a Health Canada licence for SureTune(TM)3 software for deep brain stimulation (DBS). The latest innovations in the...
-
May 31, 2017
DUBLIN - May 31, 2017 - Medtronic plc (NYSE: MDT) recently launched PRODIGY, a 1,650-patient global study, to identify individuals at high risk for opioid induced respiratory depression (OIRD), a...
-
May 18, 2017First-Ever Study on 2.0 mm DES, EuroPCR Late-Breaking Data Show Exceptional Deliverability and No Stent Thrombosis for the Resolute Onyx DES
DUBLIN and PARIS- May 18, 2017 - Medtronic plc (NYSE: MDT) today announced that the Resolute Onyx(TM) Drug-Eluting Stent (DES) met its primary endpoint of Target Lesion Failure (TLF) at one year...
-
Apr 20, 2017
DUBLIN - April 20, 2017 - Medtronic plc (NYSE: MDT) announced its sponsorship of the 23rdParkinson's Unity Walk which will be held on April 22, 2017, in New York City. Beginning at 8:30 a.m. EDT,...
-
Apr 13, 2017Data from Garmin vívofit® Series Now Syncs to Medtronic Care Management Services NetResponse® Mobile Application to Help Track Patient Health at Home
DUBLIN and OLATHE, Kan. - April 13, 2017 - Medtronic plc (NYSE:MDT), the global leader in medical technology, and Garmin International Inc., a unit of Garmin Ltd. (NASDAQ: GRMN), today announced...
-
Feb 1, 2017System First Used Commercially in U.S. This Week
DUBLIN - Feb. 1, 2017 - Medtronic plc (NYSE: MDT) has received U.S. Food and Drug Administration (FDA) 510(k) clearance for the CardioInsight(TM) Noninvasive 3D Mapping System. The CardioInsight...
-
Jan 24, 2017Expands Treatment Options for Patients in Europe with Peripheral Artery Disease Both Above and Below the Knee
DUBLIN - Jan. 24, 2017 - Medtronic plc (NYSE: MDT) today announced CE (Conformité Européene) Mark approval for the HawkOne(TM) directional atherectomy system in a lower profile size for treating...
-
Nov 29, 2016New Agreement Focuses on Improved System Efficiency at University Hospitals Cleveland Medical Center
DUBLIN and CLEVELAND - Nov. 29, 2016 - Medtronic plc (NYSE:MDT), the global leader in medical technology, today announced the signing of its first Integrated Health Solutions agreement in the...
-
Oct 24, 2016New Technology Expands Treatment Options for Patients with Peripheral Artery Disease Both Above and Below the Knee
DUBLIN - Oct. 24, 2016 - Medtronic plc (NYSE: MDT) has received U.S. Food and Drug Administration (FDA) 510(k) clearance for the HawkOne(TM) Directional Atherectomy System in a new size for...
-
Sep 20, 2016New Clinical Data Presented in Complicated Type B Aortic Dissection at VIVA 2016
DUBLIN and LAS VEGAS - Sept. 20, 2016 - Medtronic plc (NYSE: MDT) today announced new data, demonstrating safety and efficacy at three years in acute complicated Type B aortic dissection patients...
-
Aug 8, 2016New Visual-Based Platform Helps Physicians Make Informed Programming Decisions by Providing an Intuitive Visualization of Patient-Specific Images and Data
DUBLIN - August 8, 2016 - Medtronic plc (NYSE: MDT) today announced it has received CE (Conformité Européenne) Mark for SureTune2(TM) software, which provides patient-specific visualization to...
-
Jul 28, 2016- Global revenue increased approximately 25% from first quarter of 2016
HeartWare International, Inc. (NASDAQ: HTWR), a leading innovator of less-invasive, miniaturized circulatory support technologies that are revolutionizing the treatment of advanced heart failure,...
-
Jul 20, 2016Clearance Gives Spine Surgeons a New Treatment Option for Spinal Tumor Patients
DUBLIN - July 20, 2016 - Medtronic plc (NYSE: MDT) today announced U.S. Food and Drug Administration (FDA) 510(k) clearance of the CD Horizon® Fenestrated Screw Set, which can be used for...
-
Jul 11, 2016Clinical Study Conducted on Revision Subjects Support Expanded Indication for Patients with Previously Surgically Altered Tissue
DUBLIN - July 11, 2016 - Medtronic plc (NYSE: MDT) today announced U.S. Food and Drug Administration (FDA) clearance of NuVent(TM), an EM sinus dilation system, for patients with scarred,...
-
Jun 27, 2016Extends Clinical and Commercial Leadership Into Growing Circulatory Support Sector
DUBLIN and FRAMINGHAM, Mass. - June 27, 2016 - Medtronic plc (NYSE: MDT), the global leader in medical technology, and HeartWare International, Inc. (NASDAQ: HTWR), a leading innovator of...
-
Jun 7, 2016
DUBLIN - June 7, 2016 - Medtronic plc (NYSE:MDT), the global leader in medical technology, today announced its schedule of notable sessions that will be presented at the 76th Scientific Sessions...
-
Jun 1, 2016
HeartWare International, Inc. (NASDAQ:HTWR), a leading innovator of less-invasive, miniaturized circulatory support technologies that are revolutionizing the treatment of advanced heart failure,...
-
May 31, 2016New Asia Pacific Regional Headquarters to Meet Healthcare Demands in the Region
Establish Centre of Excellence with SingHealth to Enhance Patient Care for Diabetes SINGAPORE - May 31, 2016 - Medtronic plc (NYSE:MDT), a global leader in medical technology, services and...
-
May 25, 2016
DUBLIN and EINDHOVEN / ZEIST, THE NETHERLANDS - May 25, 2016 - The Nederlandse Obesitas Kliniek ('Dutch Obesity Clinic', NOK) and Medtronic plc (NYSE: MDT), a global leader in medical technology,...
-
May 11, 2016Medtronic and the Vikings to Unveil "The Horn" Monument on Medtronic Plaza Outside U.S. Bank StadiumNew 38-Ton Artistic Landmark Represents the Spirit of Progress and Partnership That Together Lead Our Community to a Better Future
MINNEAPOLIS - May 11, 2016 - Medtronic plc (NYSE: MDT) and the Minnesota Vikings today announced Medtronic's contribution of a new contemporary art sculpture on Medtronic Plaza, just outside the...
-
May 6, 2016Studies Presented at Heart Rhythm 2016 Provide Encouraging Results for ICD Lead Placement Under Ribs, Outside Heart and Veins
DUBLIN and SAN FRANCISCO - May 6, 2016 - Medtronic plc (NYSE:MDT) today announced the results of several studies evaluating a novel approach to implantable cardioverter defibrillator (ICD) therapy...
-
May 5, 2016
HeartWare International, Inc. (NASDAQ:HTWR), a leading innovator of less-invasive, miniaturized circulatory support technologies that are revolutionizing the treatment of advanced heart failure,...
-
May 4, 2016-- First quarter 2016 revenue of $55 million reflects lower year-over-year clinical trial revenue and competitive dynamics --
HeartWare International, Inc. (NASDAQ: HTWR), a leading innovator of less-invasive, miniaturized circulatory support technologies that are revolutionizing the treatment of advanced heart failure,...
-
Apr 27, 2016- Clinical Trial Evaluates Less-Invasive Thoracotomy Implant Technique of the HVAD System in Patients with Advanced Heart Failure -
HeartWare International, Inc. (NASDAQ: HTWR), a leading innovator of less-invasive, miniaturized circulatory support technologies that are revolutionizing the treatment of advanced heart failure,...
-
Apr 20, 2016- HeartWare to Host Scientific Symposiums on HVAD System Waveforms and Advancements in HVAD System Outcomes -
HeartWare International, Inc. (NASDAQ: HTWR), a leading innovator of less-invasive, miniaturized circulatory support technologies that are revolutionizing the treatment of advanced heart failure,...
-
Feb 17, 2016Approval Expands Population of Individuals Who May Benefit From the Proven Therapy
DUBLIN - February 17, 2016 - Medtronic plc (NYSE: MDT) today announced U.S. Food and Drug Administration (FDA) approval of Medtronic Deep Brain Stimulation (DBS) Therapy for use in people with...
-
Dec 14, 2015New facility to manufacture Medtronic's market- leading drug-coated balloons (DCB) technology
DUBLIN- December 14, 2015 - Medtronic plc (NYSE:MDT), a global leader in medical technology, services and solutions, today announced a new manufacturing facility in Galway, Ireland that will...
-
Dec 9, 2015Updated Labeling Allows Greater Patient Access to MRI, a Critical Diagnostic Tool
DUBLIN - December 9, 2015 - Medtronic plc (NYSE: MDT) today announced that systems within its Activa® portfolio of Deep Brain Stimulation (DBS) neurostimulators have received FDA approval for...
-
Nov 16, 2015Medtronic Showcases Aortic Leadership in Innovation with Clinical Release and Pipeline at VEITH 2015New Aortic Data Demonstrates Medtronic's Commitment to Innovation
DUBLIN - November 16, 2015 - Medtronic plc (NYSE: MDT) today announced notable clinical studies and innovative technologies that will be presented at the annual VEITHsymposium, the world's premier...
-
May 7, 2015RIO 2 Study Evaluates Safety and Effectiveness of Moving the Insertion Procedure from Hospital to Office Setting
DUBLIN – May 7, 2015 – Medtronic plc (NYSE: MDT) today announced the first inoffice implant of its miniaturized cardiac monitor as part of the Medtronic Reveal LINQ In-Office 2 (RIO 2) Study....
-
Feb 4, 2015First Uses of New Medical Device Following FDA Approval Take Place at Hospitals Nationwide
DUBLIN – Feb. 4, 2015 – U.S. hospitals this week began using a new medical device from Medtronic plc (NYSE: MDT) called the IN.PACT Admiral drug-coated balloon (DCB) to treat patients with...
-
Jan 5, 2015Pivotal Study Shows New Medical Device Provides Exceptional Clinical Outcomes and Reduces Need for Costly Repeat Procedures
MINNEAPOLIS -- Jan. 5, 2015 -- Medtronic, Inc. (NYSE: MDT) announced today that the U.S. Food and Drug Administration (FDA) has approved the company's IN.PACT Admiral drug-coated balloon (DCB) for...
-
Dec 21, 2014Results of IN.PACT SFA Trial Published in Circulation
MINNEAPOLIS -- Dec. 22, 2014 -- The results of a landmark study published this month in Circulation, the world's leading cardiovascular journal, indicate that a novel medical device from...
-
Dec 8, 2014Grants Help Mark Five Years Since Diabetes Therapy Management and Education Center Opened its Doors
SAN ANTONIO, TX - December 8, 2014 - Medtronic, Inc. (NYSE: MDT) today announced that five San Antonio community organizations will receive a combined $250,000 in Health Access Grants in...
-
Nov 5, 2014Fourth-quarter net sales up 7%; Medical Devices sales up 8%
DUBLIN, Ireland--(BUSINESS WIRE)--Nov. 5, 2014-- Covidien plc (NYSE: COV) today announced financial results for the fourth quarter of fiscal 2014. Fourth-quarter net sales of $2.73 billion...
-
Nov 4, 2014Employee Engagement, Philanthropy Among Top Reasons for Selection
BRAMPTON, ONTARIO - November 4, 2014 - Medtronic of Canada Ltd., a trusted Canadian leader in innovative medical technologies and health system solutions, is pleased to have been recognized as one...
-
Oct 7, 2014FDA-Approved System Provides Patients with Added Benefits While Testing Innovative Therapy for Bladder and Bowel Incontinence
MINNEAPOLIS - October 7, 2014 - After living with overactive bladder and urinary retention symptoms for 11 years, Jennifer LaForest, a 26-year-old woman from Auburn Hills, Michigan, recently...
-
Sep 16, 2014New Data from the OpT2mise Efficacy Study to be Presented at EASD Annual Meeting
VIENNA - September 16, 2014 - Medtronic, Inc. (NYSE:MDT) today announced that new results from the OpT2mise trial are being presented at the European Association for the Study of Diabetes (EASD) 50th
-
Aug 26, 2014Sapiens' Deep Brain Stimulation Technologies Strengthen Medtronic's Neuromodulation Portfolio and Neuroscience Leadership Position
MINNEAPOLIS and EINDHOVEN - Aug. 26, 2014 - Medtronic, Inc. (NYSE: MDT) today announced that it has acquired Sapiens Steering Brain Stimulation (Sapiens SBS), a privately held developer of deep...
-
Aug 20, 2014Facility offers educational resources, research capabilities
SÃO PAULO--(BUSINESS WIRE)--Aug. 20, 2014-- Covidien plc (NYSE: COV) today officially opened its Covidien Center of Innovation Brazil (CCI Brazil), further expanding its educational and research...
-
Aug 1, 2014System Offers Innovative Technology for Sinus Surgery. Chronic Sinusitis Affects Nearly 29 Million US Adults
MINNEAPOLIS - August 1, 2014 - Medtronic, Inc. (NYSE: MDT) today announced the launch of the NuVent(TM) EM Sinus Dilation System for the Fusion® ENT Navigation System, developed and manufactured...
-
Jul 25, 2014Net sales up 4%; Medical Devices sales up 5%
DUBLIN, Ireland--(BUSINESS WIRE)--Jul. 25, 2014-- Covidien plc (NYSE: COV) today announced financial results for the third quarter of fiscal 2014. Third-quarter net sales of $2.69 billion...
-
Jul 16, 2014Emprint™ Ablation System enables more certainty regardless of target location or tissue type
DUBLIN, Ireland--(BUSINESS WIRE)--Jul. 16, 2014-- Overcoming a significant roadblock to predictable ablation of soft tissue, Covidien plc (NYSE:COV) has unveiled an advanced ablation system that...
-
Jun 11, 2014Shareholders Invited to Attend Annual Meeting on Aug. 21, 2014
MINNEAPOLIS - June 11, 2014 - The board of directors of Medtronic, Inc. (NYSE: MDT), announced that the company's annual meeting of shareholders will be held at 10:30 a.m. CDT on Thursday, Aug....