| Su | Mo | Tu | We | Th | Fr | Sa |
|---|---|---|---|---|---|---|
| Su | Mo | Tu | We | Th | Fr | Sa |
|---|---|---|---|---|---|---|
-
Feb 18, 2025
Delivering durable revenue growth with strong earnings power; strength in Pulsed Field Ablation, Pacing, Structural Heart, Diabetes, and Neuromodulation GALWAY, Ireland, Feb. 18, 2025 /PRNewswire/...
-
Nov 19, 2024
Delivering on commitments, executing ahead of expectations, and raising guidance Innovation driving sustained growth across many franchises: TAVR, PFA, Leadless Pacemakers, Diabetes, Spine, and...
-
Aug 20, 2024
Product innovation driving growth across diversified health tech portfolio, including Automated Insulin Delivery, Transcatheter Aortic Valve Replacement, Pulsed Field Cardiac Ablation, Pain...
-
May 23, 2024
Broad-based, durable growth across the company, including Cranial & Spinal Technologies, Diabetes, Cardiac Pacing, Surgical, and Structural Heart; gaining momentum as company enters new product...
-
Feb 20, 2024
Delivers on commitments with strong growth in Core Spine, Cardiac Surgery, Structural Heart, Cardiac Pacing, and across many international markets; Diabetes increases double digits as U.S....
-
Feb 21, 2023
Delivers top and bottom line ahead of expectations, with strength in Cardiovascular and Neuroscience portfolios DUBLIN, Feb. 21, 2023 /PRNewswire/ -- Medtronic plc (NYSE:MDT) today announced...
-
Mar 24, 2022
TITAN 2 pivotal study to evaluate implantable tibial neuromodulation (TNM) device to help expand patient access to advanced therapy DUBLIN, March 24, 2022 /PRNewswire/ -- Medtronic plc (NYSE:...
-
Feb 22, 2022- New InterStim X™ system provides 10 to 15 years of battery life without the need to recharge
Medtronic plc (NYSE:MDT), a global leader in healthcare technology, today announced it received approval from the U.S. Food and Drug Administration (FDA) for InterStim X™ ― the next generation...
-
Feb 22, 2022
Company advanced its pipeline, launched new products, won share, and delivered earnings growth despite COVID-19 impact on healthcare procedure volumes DUBLIN, Feb. 22, 2022 /PRNewswire/ --...
-
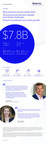
Nov 23, 2021Company continued to launch new products, win share, and deliver strong earnings growth; market procedure volumes impacted by COVID-19 resurgence
Medtronic plc (NYSE:MDT) today announced financial results for its second quarter of fiscal year 2022, which ended October 29, 2021. Key Highlights Revenue of $7.8 billion increased 3% reported...
-
Sep 22, 2021Medtronic Will Move to Request Litigation Proceedings Resume
Medtronic (NYSE:MDT), the global leader in medical technology, today announced that the Patent Trial and Appeal Board (PTAB) of the U.S. Patent and Trademark Office has affirmed an additional...
-
Sep 13, 2021Medtronic Receives Affirmation of Three Additional Sacral Neuromodulation Patents
Medtronic plc (NYSE:MDT), the global leader in medical technology, today announced it has won three additional important victories in a dispute over its intellectual property (IP) for its sacral...
-
Aug 24, 2021Strong Financial Performance on Continued Procedure Volume Recovery and Share Gains from New Product Launches
Medtronic plc (NYSE:MDT) today announced financial results for its first quarter of fiscal year 2022, which ended July 30, 2021. Key Highlights Revenue of $8.0 Billion Increased 23% Reported and...
-
May 27, 2021- Q4 Revenue of $8.2 Billion Increased 37% Reported and 32% Organic
Medtronic plc (NYSE:MDT) today announced financial results for its fourth quarter and fiscal year 2021, which ended April 30, 2021. The company reported fourth quarter worldwide revenue of $8.188...
-
Apr 27, 2021New Investigational Device Aims to Expand Access to Therapy for Patients with Incontinence; Underscores Medtronic's Leadership as Only Company to Offer Neuromodulation Options for Incontinence
Medtronic plc (NYSE:MDT), the global leader in medical technology, today announced approval from the U.S. Food and Drug Administration (FDA) to proceed with an investigational device exemption...
-
Feb 18, 2021
Medtronic plc (NYSE:MDT), the global leader in medical technology, today announced it has received approval from the United States Food and Drug Administration (FDA) for expanded MRI labeling of...
-
Oct 14, 2020
Company Raises Revenue Growth Outlook Highlights Deep and Broad Pipeline and Technology Platforms DUBLIN, Oct. 14, 2020 /PRNewswire/ -- Medtronic plc (NYSE: MDT), the global leader in medical...
-
Oct 6, 2020ELITE is the Only Study of a Rechargeable Sacral Neurostimulation System to Include All Four Indications, Including Fecal Incontinence, Which Affects Millions
Medtronic plc (NYSE:MDT), the global leader in medical technology, today announced the first patient was implanted in its ELITE study of the InterStim™ Micro system — the only study of a...
-
Sep 25, 2020U.S. Patent and Trademark Office Rejects IPR Challenge to Medtronic Patent
Medtronic (NYSE:MDT) has won an important victory in a patent dispute over intellectual property for its sacral neuromodulation device family, the InterStim™ systems. The Patent Trial and Appeal...
-
Aug 20, 2020ELITE Study Designed to Confirm Long-Term Safety, Efficacy and Patient Benefit of Sacral Neuromodulation with the InterStim™ Micro System for Treatment of Overactive Bladder, Fecal Incontinence and Non-Obstructive Urinary Retention
DUBLIN, Aug. 20, 2020 (GLOBE NEWSWIRE) -- Medtronic plc (NYSE:MDT), the global leader in medical technology, today announced it has begun recruitment of the prospective, multicenter, global,...
-
Aug 5, 2020First Patient Receives New Sacral Neuromodulation Device Implant at Cleveland Clinic
DUBLIN, Aug. 05, 2020 (GLOBE NEWSWIRE) -- Medtronic plc (NYSE:MDT), the global leader in medical technology, today announced that its recently FDA-approved InterStim™ Micro neurostimulator for...
-
Aug 3, 2020World’s Smallest Rechargeable Bladder/Bowel Control System — InterStim Micro — Features Trusted MRI Technology for Full-Body 1.5 and 3 Tesla MRI Scans
DUBLIN, Aug. 03, 2020 (GLOBE NEWSWIRE) -- Medtronic plc (NYSE:MDT), the global leader in medical technology, today announced it has received approval from the United States Food and Drug...
-
Jan 13, 2020Full-Body MRI-Conditional InterStim II and InterStim Micro Systems Offer Lifestyle-Friendly Choices in Sacral Neuromodulation Therapy to Europeans Suffering from Incontinence
DUBLIN, Jan. 13, 2020 (GLOBE NEWSWIRE) -- Medtronic plc (NYSE:MDT) today announced it has received CE Mark for its InterStim™ Micro neurostimulator and InterStim™ SureScan™ MRI leads —...
-
Nov 19, 2019Revenue of $7.7 Billion Increased 3.0% Reported and 4.1% Organic
DUBLIN, Nov. 19, 2019 (GLOBE NEWSWIRE) -- Medtronic plc (NYSE:MDT) today announced financial results for its second quarter of fiscal year 2020, which ended October 25, 2019. The company reported...
-
Nov 4, 2019Action Seeks Relief to Prevent Further Infringement of Medtronic Patents
DUBLIN, Nov. 04, 2019 (GLOBE NEWSWIRE) -- Medtronic, Inc. (NYSE:MDT) today filed a lawsuit against Axonics Modulation Technologies, Inc. (“Axonics”) alleging infringement of patents related to...